Call: 08045800468
Pharma HVAC Filter Management And Traceability System
MOQ : 1 Unit
Pharma HVAC Filter Management And Traceability System Specification
- Capacity (Person)
- Unlimited user configuration
- Language Support
- English
- Interface Type
- Web-based Dashboard, RFID/Barcode Scanning
- Usage
- Filter installation, Life-cycle monitoring, Performance validation
- Compatible System
- Central HVAC System, BMS Integration
- Security Features
- Multi-level user authentication, Encrypted data storage, Role-based access control
- Support System
- Windows, Cloud-based Backup, Real-time Technical Assistance
- Type
- Software solution for traceability and management
- Application
- Pharmaceutical HVAC Filter Management, Cleanroom Audit, Maintenance Tracking, Regulatory Compliance
- Integration Ability
- ERP and SCADA system connectivity
- Maintenance Scheduler
- Integrated filter service calendar
- Scalability
- Supports multi-facility operations
- Reports & Analytics
- Customizable filter status and compliance reports
- User Management
- Hierarchical permission levels for supervisor and staff
- Data Export
- CSV, PDF, and secure API for third-party integration
- Operating Temperature
- 0C to 50C (hardware components only)
- Alert System
- Scheduled filter replacement reminders and automated notifications
- Data Logging
- Automated event recording and change history
- Installation Mode
- On-premises or SaaS (cloud-based)
- Mobile Compatibility
- Android and iOS support for field operations
- Power Supply
- Operates on standard 220V/50Hz AC supply (for associated hardware if any)
- Storage Capacity
- Cloud infrastructure scalable based on user needs
- Regulatory Standards
- Compliant with GMP, FDA 21 CFR Part 11, and ISO 14644
Pharma HVAC Filter Management And Traceability System Trade Information
- Minimum Order Quantity
- 1 Unit
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 10 Units Per Month
- Delivery Time
- 30 Days
- Main Domestic Market
- All India
About Pharma HVAC Filter Management And Traceability System
Reserve your edge in pharmaceutical facility management with our Pharma HVAC Filter Management and Traceability System. Take advantage of automated event recording, customizable analytics, and breathtaking scalability all in one ace package. Real-time mobile support, a monumental alert system, and seamless data export enhance compliance and traceability. Designed for multi-facility operations, it ensures cloud-backed security, effortless integration with ERP/SCADA, and streamlined maintenance. Experience user-focused features, advanced regulatory adherence, hierarchical user management, and robust hardware compatibilities-making this solution indispensable for manufacturers and service providers dedicated to excellence in cleanroom performance.
Precision Material Management for Critical Environments
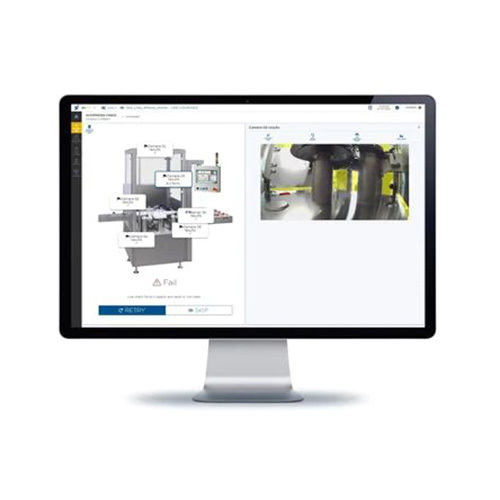
This system is expertly crafted for traceable filter handling in pharmaceuticals, delivering ace performance for cleanroom audits and compliant maintenance. Suitable for use in manufacturing, research, and service provider environments, it is the ultimate software solution for real-time filter installation tracking, life-cycle monitoring, and performance validation. Its flexibility allows seamless integration with central HVAC and BMS systems. With robust web-based dashboards, RFID and barcode scanning support, it offers effortless use for unlimited personnel.
Dependable Supply, Sample Access & Secure Packaging
Our Pharma HVAC Filter Management and Traceability System is dispatched promptly upon order confirmation, with the supply ability to meet high-volume, multi-facility requirements. Clients can request a sample to evaluate its interface and features before making a monumental decision. Every package is securely prepared, ensuring the software and associated hardware charge safely reaches you. Each shipment is backed with technical assistance for smooth implementation, whether delivered as on-premises or SaaS, locally or nationwide.
Precision Material Management for Critical Environments
This system is expertly crafted for traceable filter handling in pharmaceuticals, delivering ace performance for cleanroom audits and compliant maintenance. Suitable for use in manufacturing, research, and service provider environments, it is the ultimate software solution for real-time filter installation tracking, life-cycle monitoring, and performance validation. Its flexibility allows seamless integration with central HVAC and BMS systems. With robust web-based dashboards, RFID and barcode scanning support, it offers effortless use for unlimited personnel.
Dependable Supply, Sample Access & Secure Packaging
Our Pharma HVAC Filter Management and Traceability System is dispatched promptly upon order confirmation, with the supply ability to meet high-volume, multi-facility requirements. Clients can request a sample to evaluate its interface and features before making a monumental decision. Every package is securely prepared, ensuring the software and associated hardware charge safely reaches you. Each shipment is backed with technical assistance for smooth implementation, whether delivered as on-premises or SaaS, locally or nationwide.
FAQ's of Pharma HVAC Filter Management And Traceability System:
Q: How does the system enhance compliance with regulatory standards?
A: The system aligns with GMP, FDA 21 CFR Part 11, and ISO 14644, offering automated event recording, traceable maintenance history, and customizable compliance reports, helping users meet regulatory obligations efficiently.Q: What kind of organisations can take advantage of this system?
A: It is designed for pharmaceutical manufacturers, service providers, and suppliers, supporting operations ranging from single facilities to large-scale, multi-location enterprises across India.Q: When will I receive scheduled filter replacement notifications?
A: Automated notifications and reminders are sent based on the integrated maintenance scheduler, helping you reserve time for crucial filter replacements before performance is compromised.Q: Where can the system be integrated within existing infrastructure?
A: Compatible with central HVAC systems, BMS, and ERP/SCADA solutions, the system can be installed on-premises or used as a cloud-based (SaaS) service for optimal versatility.Q: What packaging and support options are provided upon dispatch?
A: Each order is securely packaged, and technical support is charged with assisting your team in implementation. Cloud-based backup and real-time help are included.Q: How does user management and data security operate in the system?
A: Hierarchical permissions, multi-level user authentication, encrypted storage, and role-based access control work together to create a secure, compliant environment for all personnel.
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Pharma Compliance And Traceability Systems Category
Digital Line Clearance System
Minimum Order Quantity : 1 Unit
Support System : Windows OS, Custom embedded systems
Application : Pharmaceutical production lines, Laboratory environments, Electronic manufacturing, Biotechnology setups
Security Features : User authentication, Access control, Digital signature, Encrypted data logs
Compatible System : SCADA, MES, Pharmaceutical ERP systems
Capacity (Person) : Multiuser (supports 1050 simultaneous users)

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry